Nitrogen Rejection
After the Natural Gas Liquids (NGLs) have been recovered, the dry gas enters a nitrogen rejection unit. The nitrogen rejection unit serves to remove the inert nitrogen gas mainly to improve the quality of the natural gas product. A large amount of nitrogen gas can decrease the heating value of the natural gas. This unit may be comprised of a cryogenic, adsorbent, or membrane processing technology. The cryogenic distillation method that involves lowering the temperature of natural gas is similar to the discussion in the Hydrocarbon Recovery module.
Adsorption
This process is a much simpler batch operation compared to cryogenic distillation. A pressure swing adsorption is typically used in nitrogen removal units that can handle 2 to 15 MMscfd (Million standard cubic feet per day). The means of adsorption depends on the difference in adsorbate polarity and size of the adsorbent. The two steps in regenerative adsorption separation are adsorption to separate the species, and desorption of adsorbate to prepare the adsorbent for further use. Ideally, nitrogen will be the adsorbate so the content of natural gas leaving the adsorbent bed will have a reduced nitrogen content.
Carbon molecular sieves (CMS) may be used in pressure swing adsorption that is prepared by the processing of coal or wood carbon. Nitrogen in the feed entering the adsorbing bed becomes adsorbed until the outlet concentration increases due to bed breakthrough. At that stage, the bed will be switched into a regeneration mode and the feed will be switched to a regenerated bed from a previous cycle to continue the batch operation.
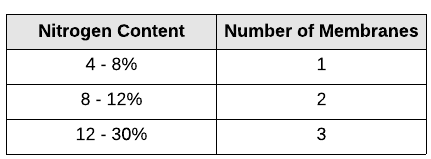
Membrane Processing
This method is used for nitrogen content between 4 to 30% to be considered economically feasible. The number of membranes used in a single unit operation depends on the nitrogen content of the gas which is simplified in the table below.
Membranes depend on the permeability of the selected gas being permeated and the selectivity between the permeate and retentate. The basic membrane concept is illustrated further in the Membranes module. The membrane may be methane selective or nitrogen selective, but research has shown methane selective membranes to produce a better quality of natural gas to pipelines.
Sulfur Recovery
Sulfur that has been removed from the acid gas removal unit can either be disposed of in gas form by injecting it into underground formations or be converted into a usable product which is elemental sulfur. The Claus process is the most commonly used method to convert hydrogen sulfide to elemental sulfur. Additional treatment is required, as the product produced by this process does not meet the environmental emissions requirements.
Claus Process
H 2 S + (3/2) O 2 ⇆ H 2 O + SO 2
2H 2 S + SO 2 ⇆ H 2 O + (3/x) S x
The hydrogen sulfide from acid gas removal is mixed with air using a mixer and enters a furnace at 600 ° F, where a catalytic combustion reaction occurs. High-pressure steam is released from this exothermic process, and sulfur is formed in the vapor phase. Sulfur vapor exists as S x, where x can be from 1 to 8. The sulfur extracted from the reaction passes through a series of catalytic reactors. The mixture from the reactors shifts the equilibrium of the sequential combustion reaction shown above to produce more sulfur. The sulfur vapor is then condensed in a condenser using water as a cooling medium.
Acknowledgments
- Bertsch Holding GmbH
References
- Kidnay, A.; Parrish, W. Fundamentals of Natural Gas Processing. Boca Raton: CRC Press, 2006
- Engineering data book: volumes I & II: SI version; Gas Processors Suppliers Association: Tusla, OK, 2012.
- Mokhatab, S.; Poe, W. A.; & Mak, J. Y. Handbook of Natural Gas Transmission and Processing. Boston: Gulf Professional Publishing, 2015, 191-217
- Carroll, J. Natural Gas Hydrates – A Guide for Engineers; 3rd ed.; Elsevier, 2018.
Developers
- Nuramani Saiyidah Binti Ramli
- James Rivard